|
Toll Free: (877) 796-6397 -- USA and Canada only -- |
Fax: +1-832-582-8590 Orders: +1-832-582-8158 |
Tech Support: +1-832-582-8158 Ext:3 Please provide your Order Number in the email. |
Technical Data
| Formula | C40H53N7O5S2 |
|||
| Molecular Weight | 776.02 | CAS No. | 1004316-88-4 | |
| Solubility (25°C)* | In vitro | DMSO | 100 mg/mL (128.86 mM) | |
| Ethanol | 100 mg/mL (128.86 mM) | |||
| Water | Insoluble | |||
|
* <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. * Room temperature shipping (Stability testing shows this product can be shipped without any cooling measures.) |
||||
Preparing Stock Solutions
Biological Activity
| Description | Cobicistat is a potent and selective inhibitor of CYP3A with IC50 of 30-285 nM. | ||
|---|---|---|---|
| Targets |
|
||
| In vitro | Cobicistat (GS-9350) is a potent, and selective inhibitor of human cytochrome P450 3A (CYP3A) enzymes as a pharmacoenhancer. GS-9350 inhibits CYP3A with IC50 spectrum from 30 nM to 285 nM. In contrast to ritonavir, GS-9350 is devoid of anti-HIV activity, with IC50 of > 30μM against HIV-1 protease and EC50 of > 30μM in MT-2 HIV infection assay, and is thus more suitable for use in boosting anti-HIV drugs without risking selection of potential drug-resistant HIV variants. GS-9350 shows reduced liability for drug interactions and may have potential improvements in tolerability over ritonavir. [1] |
Protocol (from reference)
| Kinase Assay:[1] |
|
|---|
Customer Product Validation
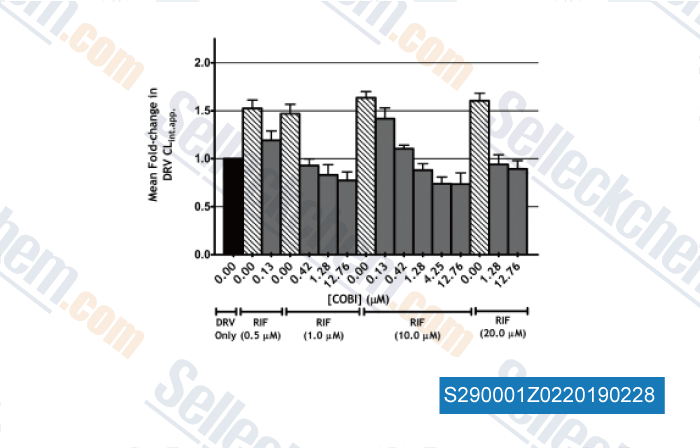
-
Data from [Data independently produced by , , Antimicrob Agents Chem, 2017, doi:10.1128/AAC.01776-16]
Selleck's Cobicistat has been cited by 11 publications
| Establishment and Characterization of NCC-PMP1-C1: A Novel Patient-Derived Cell Line of Metastatic Pseudomyxoma Peritonei [ J Pers Med, 2022, 12(2)258] | PubMed: 35207746 |
| Establishment and characterization of NCC-UPS4-C1: a novel cell line of undifferentiated pleomorphic sarcoma from a patient with Li-Fraumeni syndrome [ Hum Cell, 2022, 10.1007/s13577-022-00671-y] | PubMed: 35118583 |
| Establishment and characterization of NCC-MFS4-C1: a novel patient-derived cell line of myxofibrosarcoma [ Hum Cell, 2021, 34(6):1911-1918] | PubMed: 34383271 |
| Establishment and characterization of the NCC-GCTB4-C1 cell line: a novel patient-derived cell line from giant cell tumor of bone [ Hum Cell, 2021, 10.1007/s13577-021-00639-4] | PubMed: 34731453 |
| Establishment and characterization of novel patient-derived cell lines from giant cell tumor of bone [ Hum Cell, 2021, 10.1007/s13577-021-00579-z] | PubMed: 34304386 |
| Establishment and characterization of patient-derived cancer models of malignant peripheral nerve sheath tumors. [ Cancer Cell Int, 2020, 19;20:58] | PubMed: 32099531 |
| Establishment and characterization of NCC-MFS2-C1: a novel patient-derived cancer cell line of myxofibrosarcoma [ Hum Cell, 2020, 10.1007/s13577-020-00420-z] | PubMed: 32870449 |
| Establishment and characterization of a novel cell line, NCC-TGCT1-C1, derived from a patient with tenosynovial giant cell tumor [ Hum Cell, 2020, 10.1007/s13577-020-00425-8] | PubMed: 32886306 |
| Human Platelet Dysregulation by Antiretroviral Drugs and Cigarette Smokein vitro [ ProQuest, 2019, 13863770] | PubMed: None |
| Interaction of Rifampin and Darunavir-Ritonavir or Darunavir-Cobicistat In Vitro. [ Antimicrob Agents Chemother, 2017, 61(5)] | PubMed: 28193650 |
RETURN POLICY
Selleck Chemical’s Unconditional Return Policy ensures a smooth online shopping experience for our customers. If you are in any way unsatisfied with your purchase, you may return any item(s) within 7 days of receiving it. In the event of product quality issues, either protocol related or product related problems, you may return any item(s) within 365 days from the original purchase date. Please follow the instructions below when returning products.
SHIPPING AND STORAGE
Selleck products are transported at room temperature. If you receive the product at room temperature, please rest assured, the Selleck Quality Inspection Department has conducted experiments to verify that the normal temperature placement of one month will not affect the biological activity of powder products. After collecting, please store the product according to the requirements described in the datasheet. Most Selleck products are stable under the recommended conditions.
NOT FOR HUMAN, VETERINARY DIAGNOSTIC OR THERAPEUTIC USE.
