|
How to Cite 1. For In-Text Citation (Materials & Methods): 2. For Key Resources Table: |
||
|
Toll Free: (877) 796-6397 -- USA and Canada only -- |
Fax: +1-832-582-8590 Orders: +1-832-582-8158 |
Tech Support: +1-832-582-8158 Ext:3 Please provide your Order Number in the email. We strive to reply to |
Technical Data
| Formula | C33H38N4O6 |
||||||||||||||
| Molecular Weight | 586.68 | CAS No. | 97682-44-5 | ||||||||||||
| Solubility (25°C)* | In vitro | DMSO | 25 mg/mL (42.61 mM) | ||||||||||||
| Water | Insoluble | ||||||||||||||
| Ethanol | Insoluble | ||||||||||||||
| In vivo (Add solvents to the product individually and in order) |
|
||||||||||||||
|
* <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. * Room temperature shipping (Stability testing shows this product can be shipped without any cooling measures.) |
|||||||||||||||
Preparing Stock Solutions
Biological Activity
| Description | Irinotecan is a topoisomerase I inhibitor for LoVo cells and HT-29 cells with IC50 of 15.8 μM and 5.17 μM, respectively. | |
|---|---|---|
| Targets |
|
|
| In vitro | Irinotecan is activated to SN-38 by carboxylesterases to become able to interact with its target, topoisomerase I. This compound induces similar amounts of cleavable complexes at its IC50 in LoVo cells and HT-29 cell lines. SN-38 induces a concentration-dependent formation of cleavable complexes, which is not significantly different in LoVo cells and HT-29 cell lines. Cell accumulation of this chemical is markedly different, reaching consistently higher levels in HT-29 cells than in LoVo cells. The lactone E-ring of this compound and SN-38 hydrolyses reversibly in aqueous solutions, and the interconversion between the lactone and carboxylate forms is dependent on pH and temperature. Liver is primarily responsible for the activation of this agent to SN-38. At equal concentrations of this drug and SN-38 glucuronide, the rate of beta-glucuronidase-mediated SN-38 production is higher than that formed from this compound in both tumour and normal tissue. It is also converted to SN-38 in intestines, plasma and tumor tissues. This agent is significantly more active in SCLC than in NSCLC cell lines, whereas no significant difference between histological types is observed with SN-38. | |
| In vivo | In COLO 320 xenografts, Irinotecan induces a maximum growth inhibition of 92%. A single dose of this compound significantly increases amounts of topoisomerase I covalently bound to DNA in stomach, duodenum, colon and liver. Concomitantly, the Irinotecan-treated group shows significantly higher amounts of DNA strand breaks in colon mucosa cells compared to the control group. | |
| Features | Irinotecan is a prodrug that is used to treat metastatic colorectal cancer. |
Protocol (from reference)
| Cell Assay: |
|
|---|---|
| Animal Study: |
|
References
|
Customer Product Validation
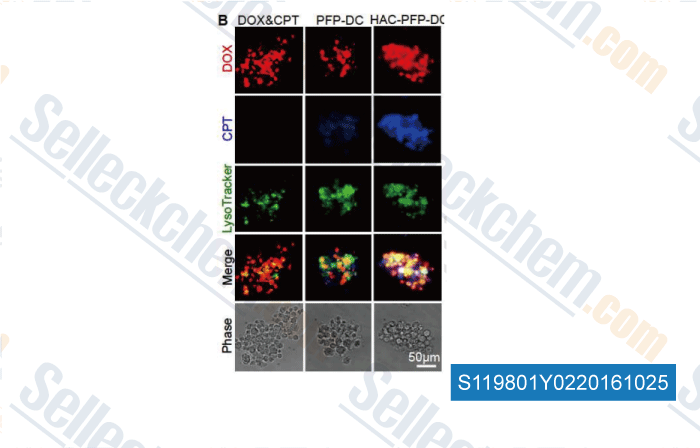
-
, , Biomaterials, 2015, 72:74-89.
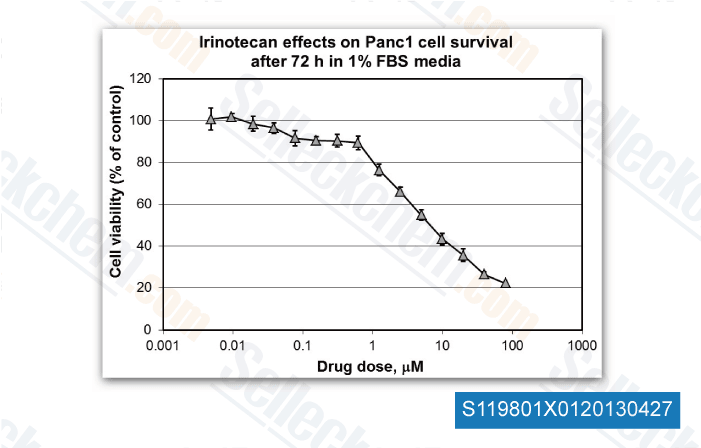
-
Dr. Edita Aksamitiene from Thomas Jefferson University, , Dr. Mikhail Menshikov of Cardiology Research Center
Selleck's Irinotecan (CPT-11) Has Been Cited by 57 Publications
| A pancreatic cancer organoid biobank links multi-omics signatures to therapeutic response and clinical evaluation of statin combination therapy [ Cell Stem Cell, 2025, S1934-5909(25)00265-6] | PubMed: 40812300 |
| A patient-derived T cell lymphoma biorepository uncovers pathogenetic mechanisms and host-related therapeutic vulnerabilities [ Cell Rep Med, 2025, S2666-3791(25)00102-8] | PubMed: 40147445 |
| Targeting of the G9a, DNMT1 and UHRF1 epigenetic complex as an effective strategy against pancreatic ductal adenocarcinoma [ J Exp Clin Cancer Res, 2025, 44(1):13] | PubMed: 39810240 |
| A Novel Modulator of Resistance for Oxaliplatin-Based Therapy for Colorectal Cancer: The ESCRT Family Member VPS4A [ Cells, 2025, 14(12)929] | PubMed: 40558556 |
| Homologous Repair-Deficient Pancreatic Cancer: Refined Targeting of DNA Damage Response is an Effective Therapeutic Strategy [ United Eur Gastroent, 2025, 13(7):1328-1342] | PubMed: 40823818 |
| The Synergistic Effect of PARP Inhibitors and Irinotecan in Small Cell Lung Cancer Cells [ Cancer Res Treat, 2025, 10.4143/crt.2024.728] | PubMed: 39788642 |
| Proteomic profiling identifies upregulation of aurora kinases causing resistance to taxane-type chemotherapy in triple negative breast cancer [ Sci Rep, 2025, 15(1):3211] | PubMed: 39863788 |
| Species-specific in vivo exposure assessment and in vivo-in vitro correlation of the carboxylate esters prodrug HD56 targeting FK506 binding proteins: The pivotal role of humanized mice [ Drug Metab Dispos, 2025, 53(4):100049] | PubMed: 40073534 |
| Low-cost robotic manipulation of live microtissues for cancer drug testing [ Sci Adv, 2025, 11(20):eads1631] | PubMed: 40367160 |
| AMG 193, a Clinical Stage MTA-Cooperative PRMT5 Inhibitor, Drives Antitumor Activity Preclinically and in Patients With MTAP-Deleted Cancers [ Cancer Discov, 2024, 10.1158/2159-8290.CD-24-0887] | PubMed: 39282709 |
RETURN POLICY
Selleck Chemical’s Unconditional Return Policy ensures a smooth online shopping experience for our customers. If you are in any way unsatisfied with your purchase, you may return any item(s) within 7 days of receiving it. In the event of product quality issues, either protocol related or product related problems, you may return any item(s) within 365 days from the original purchase date. Please follow the instructions below when returning products.
SHIPPING AND STORAGE
Selleck products are transported at room temperature. If you receive the product at room temperature, please rest assured, the Selleck Quality Inspection Department has conducted experiments to verify that the normal temperature placement of one month will not affect the biological activity of powder products. After collecting, please store the product according to the requirements described in the datasheet. Most Selleck products are stable under the recommended conditions.
NOT FOR HUMAN, VETERINARY DIAGNOSTIC OR THERAPEUTIC USE.
