
- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Hydrocortisone
Synonyms: NSC 10483
Hydrocortisone is a steroid hormone or glucocorticoid produced by the adrenal gland.
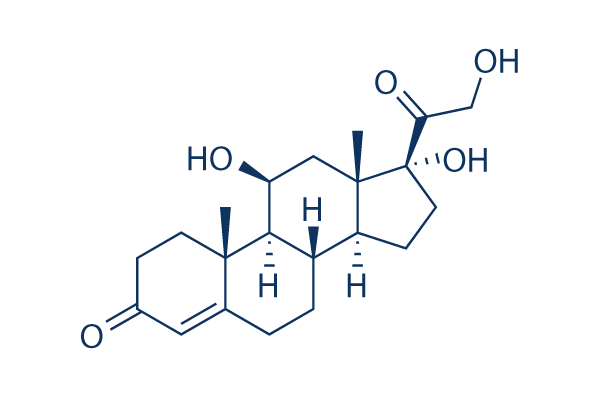
Hydrocortisone Chemical Structure
CAS: 50-23-7
Selleck's Hydrocortisone has been cited by 28 publications
Purity & Quality Control
Batch:
Purity:
99.99%
99.99
Hydrocortisone Related Products
Choose Selective Glucocorticoid Receptor Inhibitors
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| mouse RAW264.7 cells | Function assay | 24 h | Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of IFN-gamma induced NO production after 24 hrs by Griess reaction, IC50=0.061 μM | 21361338 | ||
| mouse L929 cells | Growth inhibition assay | 6 days | Growth inhibition of mouse L929 cells after 6 days | 926113 | ||
| RAW264.7 | Antiinflammatory assay | Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced NO production by MTT assay, IC50 = 40.64 μM. | 20879743 | |||
| RAW264.7 | Function assay | 24 hrs | Inhibition of LPS-induced nitric oxide production in mouse RAW264.7 cells after 24 hrs by Griess reaction based spectrophotometry, IC50 = 40.64 μM. | 21807513 | ||
| RAW264.7 | Function assay | 24 hrs | Inhibition of LPS-induced NO production in mouse RAW264.7 cells after 24 hrs by Griess reaction, IC50 = 40.64 μM. | 23067550 | ||
| RAW264.7 | Antiinflammatory assay | 24 hrs | Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced nitric oxide production after 24 hrs by Griess assay, IC50 = 43.8 μM. | 23755850 | ||
| L929 | Function assay | 20 hrs | Displacement of 1 x 10'-8 M of [1,2,3-3H]-triamcinolone acetonide from glucocorticoid receptor in soluble fraction of mouse L929 cells after 20 hrs, Kd = 0.043 μM. | 926113 | ||
| HEK293 | Function assay | Inhibition of MR-mediated transactivation of galactosidase reporter gene in HEK293 cells expressing 11betaHSD1 | 16759088 | |||
| RAW264.7 | Antiinflammatory assay | 0.5 to 50 ug/ml | 4 hrs | Antiinflammatory activity against LPS-stimulated mouse RAW264.7 cells assessed as inhibition of IL-10 production at 0.5 to 50 ug/ml pretreated for 4 hrs followed by challenge with 1 ug/mL LPS for 48 hrs by ELISA relative to control | 21384845 | |
| RAW264.7 | Antiinflammatory assay | 0.5 to 50 ug/ml | 4 hrs | Antiinflammatory activity against LPS-stimulated mouse RAW264.7 cells assessed as inhibition of IL-6 production at 0.5 to 50 ug/ml pretreated for 4 hrs followed by challenge with 1 ug/mL LPS for 48 hrs by ELISA relative to control | 21384845 | |
| RAW264.7 | Antiinflammatory assay | 0.5 to 50 ug/ml | 4 hrs | Antiinflammatory activity against LPS-stimulated mouse RAW264.7 cells assessed as inhibition of IL1-beta production at 0.5 to 50 ug/ml pretreated for 4 hrs followed by challenge with 1 ug/mL LPS for 48 hrs by ELISA relative to control | 21384845 | |
| RAW264.7 | Antiinflammatory assay | 0.5 to 50 ug/ml | 4 hrs | Antiinflammatory activity against LPS-stimulated mouse RAW264.7 cells assessed as inhibition of TNFalpha production at 0.5 to 50 ug/ml pretreated for 4 hrs followed by challenge with 1 ug/mL LPS for 48 hrs by ELISA relative to control | 21384845 | |
| RAW264.7 | Antiinflammatory assay | 0.5 to 50 ug/ml | 4 hrs | Antiinflammatory activity against LPS-stimulated mouse RAW264.7 cells assessed as inhibition of nitric oxide production at 0.5 to 50 ug/ml pretreated for 4 hrs followed by challenge with 1 ug/mL LPS for 48 hrs by Griess reaction method relative to control | 21384845 | |
| neural precursor cells | Function assay | Inhibition of neurosphere proliferation of mouse neural precursor cells by MTT assay | 17417631 | |||
| RAW264.7 | Antiinflammatory assay | 10 uM | 2 hrs | Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced increase in TNF-alpha level at 10 uM incubated for 2 hrs prior to LPS challenge measured after 22 hrs by ELISA relative to control | 25515561 | |
| RAW264.7 | Antiinflammatory assay | 10 uM | 2 hrs | Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced increase in IL-6 level at 10 uM incubated for 2 hrs prior to LPS challenge measured after 22 hrs by ELISA relative to control | 25515561 | |
| RAW264.7 | Antiinflammatory assay | 10 uM | 2 hrs | Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced increase in IL-1beta level at 10 uM incubated for 2 hrs prior to LPS challenge measured after 22 hrs by ELISA relative to control | 25515561 | |
| RAW264.7 | Antiinflammatory assay | 10 uM | 2 hrs | Antiinflammatory activity in mouse RAW264.7 cells assessed as inhibition of LPS-induced nitric oxide production at 10 uM incubated for 2 hrs prior to LPS challenge measured after 22 hrs relative to vehicle-treated control | 25515561 | |
| B16F10 | Apoptosis assay | 20 uM | 36 hrs | Induction of apoptosis in mouse B16F10 cells assessed as increase in cleaved caspase 3 level at 20 uM after 36 hrs by western blot analysis | 26695732 | |
| B16F10 | Apoptosis assay | 20 uM | 36 hrs | Induction of apoptosis in mouse B16F10 cells assessed as increase in p53 expression at 20 uM after 36 hrs by propidium iodide staining based FACS analysis | 26695732 | |
| B16F10 | Apoptosis assay | 20 uM | 36 hrs | Induction of apoptosis in mouse B16F10 cells assessed as increased Bax expression at 20 uM after 36 hrs by western blot analysis | 26695732 | |
| B16F10 | Antitumor activity assay | 6.11 mg/kg | Antitumor activity against mouse B16F10 cells xenografted in C57BL/J mouse assessed as inhibition of tumor growth at 6.11 mg/kg, ip | 26695732 | ||
| B16F10 | Apoptosis assay | 20 uM | 36 hrs | Induction of apoptosis in mouse B16F10 cells assessed as increase in cytochrome C level at 20 uM after 36 hrs by western blot analysis | 26695732 | |
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | Hydrocortisone is a steroid hormone or glucocorticoid produced by the adrenal gland. | |
|---|---|---|
| Targets |
|
| In vitro | ||||
| In vitro | Hydrocortisone prevents endothelial barrier breakdown in response to pro-inflammatory stimuli (TNFalpha administration) in the human brain microvascular endothelial cell line hCMEC/D3, which could be demonstrated to be partly based on maintenance of occludin levels. [1] Hydrocortisone-treated dendritic cells (DCs) show a decreased expression of MHC II molecules, the costimulatory molecule CD86, and the DC-specific marker CD83, as well as a strongly reduced IL-12 secretion. Hydrocortisone-treated DCs inhibits production of IFN-gamma but induces an increased release of IL-4 and no change in IL-5. Hydrocortisone reduces T-cell proliferation in dendritic cells. [2] Hydrocortisone prevents TNF-alpha induced severe degradation of the glycocalyx, increased coronary resistance, heightened vascular leak and permeability to hydroxyethyl starch and caused mast-cell degranulation in isolated guinea pig hearts. [3] Hydrocortisone reduces postischemic oxidative stress, perfusion pressure, and transudate formation in isolated guinea pig hearts. Hydrocortisone inhibits postischemic shedding of syndecan-1, heparan sulfate, and hyaluronan, as well as release of histamine from resident mast cells. [4] Hydrocortisone increases the levels of IL-4-induced germ line C epsilon transcripts by twofold and delivers the signal required for transcription of mature C epsilon mRNA. Hydrocortisone induces S mu-S epsilon deletional switch recombination in IL-4-treated B cells, and support a model of sequential isotype switching from IgM to IgE via IgG4. [5] |
|||
|---|---|---|---|---|
| Cell Research | Cell lines | CD34+ cells | ||
| Concentrations | 1 μM | |||
| Incubation Time | 16 h | |||
| Method | Cells were treated with indicated concentrations of drug. |
|||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | parkin / AIMP2 CREB |
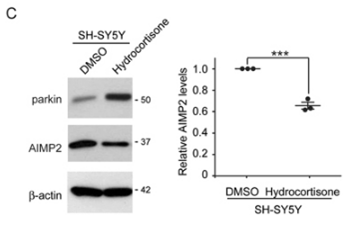
|
28366931 | |
| Immunofluorescence | Glut2 |
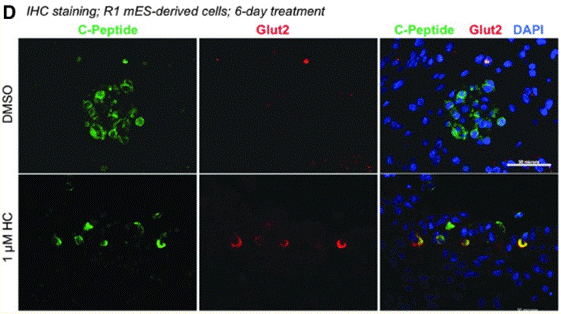
|
29717618 | |
| Growth inhibition assay | Cell viability |
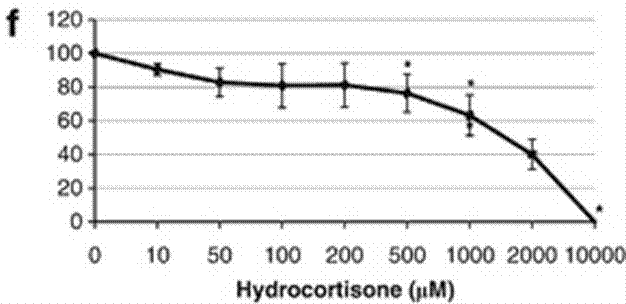
|
22829975 | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05741762 | Recruiting | Critical Illness|Adrenal Insufficiency|Septic Shock |
Dr Adnan Agha|United Arab Emirates University |
January 31 2023 | -- |
| NCT05607901 | Recruiting | Dermatologic Disease |
Tanta University |
October 28 2022 | Phase 2 |
| NCT05324618 | Completed | Atopic|Dermatitis |
Ain Shams University|National Hepatology & Tropical Medicine Research Institute |
May 15 2022 | Phase 4 |
Chemical Information & Solubility
| Molecular Weight | 362.46 | Formula | C21H30O5 |
| CAS No. | 50-23-7 | SDF | Download Hydrocortisone SDF |
| Smiles | CC12CCC(=O)C=C1CCC3C2C(CC4(C3CCC4(C(=O)CO)O)C)O | ||
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 72 mg/mL ( (198.64 mM); Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Ethanol : 15 mg/mL Water : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Tags: buy Hydrocortisone | Hydrocortisone ic50 | Hydrocortisone price | Hydrocortisone cost | Hydrocortisone solubility dmso | Hydrocortisone purchase | Hydrocortisone manufacturer | Hydrocortisone research buy | Hydrocortisone order | Hydrocortisone mouse | Hydrocortisone chemical structure | Hydrocortisone mw | Hydrocortisone molecular weight | Hydrocortisone datasheet | Hydrocortisone supplier | Hydrocortisone in vitro | Hydrocortisone cell line | Hydrocortisone concentration | Hydrocortisone nmr







































