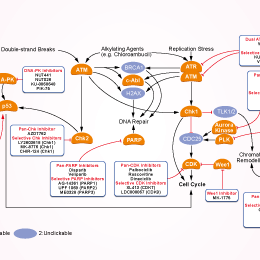
- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Oxaliplatin
Synonyms: L-OHP,NSC 266046
Oxaliplatin is a DNA alkylating agent that activates autophagy. Oxaliplatin inhibits DNA synthesis by conforming DNA adducts in RT4, TCCSUP, A2780, HT-29, U-373MG, U-87MG, SK-MEL-2, and HT-144 cells.Solutions are unstable and should be fresh-prepared.DMSO is not recommended to dissolve platinum-based drugs, which can easily lead to drug inactivation.
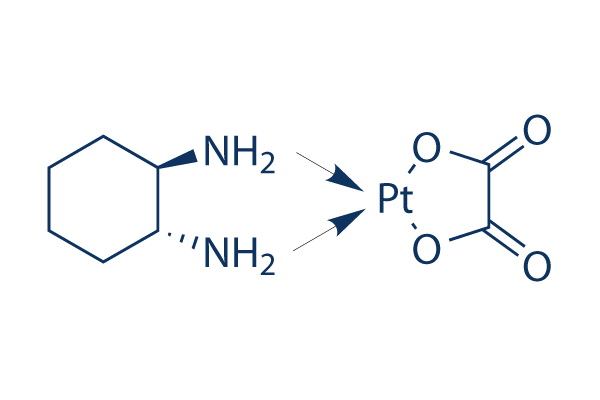
Oxaliplatin Chemical Structure
CAS: 61825-94-3
Selleck's Oxaliplatin has been cited by 222 publications
Purity & Quality Control
Batch:
Purity:
99.78%
99.78
Oxaliplatin Related Products
Signaling Pathway
Choose Selective DNA/RNA Synthesis Inhibitors
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| Caco2 | Function Assay | 30/100 μM | 16 h | DMSO | activates Nrf2 | 24556415 |
| Caco2 | Function Assay | 3/10/30 μM | 16 h | DMSO | increases the mRNA levels of AKR1C1, NQO1, HO-1, MRP2, andMRP3 dose-dependently | 24556415 |
| Caco2 | Function Assay | 30 μM | 24 h | DMSO | induces the expression of HO-1, AKR1C, and NQO1 | 24556415 |
| SW620 | Growth Inhibition Assay | 10-70 mg/L | 24/48/72 h | inhibits cell growth in both time and dose dependent manner | 24646305 | |
| SW480 | Growth Inhibition Assay | 48 h | IC50=20.8 ug/mL | 24720675 | ||
| HCT116 | Function Assay | 2/5 µM | 24/48 h | suppresses survivin mRNA expression | 24761411 | |
| HT-29 | Growth Inhibition Assay | 1 μM | 0-72 h | inhibits cell growth in a time dependent manner | 24997451 | |
| SW480 | Growth Inhibition Assay | 1 μM | 0-72 h | inhibits cell growth in a time dependent manner | 24997451 | |
| HUVEC | Growth Inhibition Assay | IC50=11.30 ± 1.02 μM | 25307448 | |||
| HEK293 | Growth Inhibition Assay | IC50=8.82 ± 5.59 μM | 25307448 | |||
| HT-29 | Growth Inhibition Assay | IC50>50 μM | 25307448 | |||
| HCT-116 | Growth Inhibition Assay | IC50=6.24 ± 2.97 μM | 25307448 | |||
| MCF-7 | Growth Inhibition Assay | IC50=14.24 ± 1.82 μM | 25307448 | |||
| HepG2 | Growth Inhibition Assay | IC50=14.24 ± 1.82 μM | 25307448 | |||
| A549 | Growth Inhibition Assay | IC50=51.08 ± 10.96 μM | 25307448 | |||
| SGC7901 | Growth Inhibition Assay | IC50=21.73 ± 3.08 μM | 25307448 | |||
| COC1 | Growth Inhibition Assay | IC50=46.20 ± 3.14 μM | 25307448 | |||
| HCT116 | Growth Inhibition Assay | 72 h | IC50=6.23±0.75 µg/mL | 25360631 | ||
| SW480 | Growth Inhibition Assay | 72 h | IC50=10.7±2.26 µg/mL | 25360631 | ||
| MIAPaCa-2 | Function Assay | 25 µM | 24/48 h | induces cleavage of PARP, caspase-9, caspase-8 and caspase-3 | 25444914 | |
| Panc-1 | Function Assay | 25 µM | 24/48 h | induces cleavage of PARP, caspase-9, caspase-8 and caspase-3 | 25444914 | |
| MIAPaCa-2 | Apoptosis Assay | 25 µM | 24 h | induces apoptosis in a synergistic manner combined with WA | 25444914 | |
| Panc-1 | Apoptosis Assay | 25 µM | 24 h | induces apoptosis in a synergistic manner combined with WA | 25444914 | |
| HPDE | Cell Viability Assay | 25/50 μM | 24/48 h | inhibits proliferation of PC cells in a synergistic manner combined with WA | 25444914 | |
| SW1990 | Cell Viability Assay | 25/50 μM | 24/48 h | inhibits proliferation of PC cells in a synergistic manner combined with WA | 25444914 | |
| MIAPaCa-2 | Cell Viability Assay | 25/50 μM | 24/48 h | inhibits proliferation of PC cells in a synergistic manner combined with WA | 25444914 | |
| Panc-1 | Cell Viability Assay | 25/50 μM | 24/48 h | inhibits proliferation of PC cells in a synergistic manner combined with WA | 25444914 | |
| A549/CDDP | Growth Inhibition Assay | IC50=18.6 ± 1.2 μM | 25625243 | |||
| A549 | Growth Inhibition Assay | IC50=5.8 ± 0.6 μM | 25625243 | |||
| SW480 | Function Assay | 10 µg/ml | 24 h | enhances cellular autophagic flux | 25749420 | |
| SW620 | Function Assay | 10 µg/ml | 24 h | enhances cellular autophagic flux | 25749420 | |
| SW480 | Function Assay | 10 µg/ml | 24 h | increases LC3-II accumulation and decreases P62 expression | 25749420 | |
| SW620 | Function Assay | 10 µg/ml | 24 h | increases LC3-II accumulation and decreases P62 expression | 25749420 | |
| HT-29 | Growth Inhibition Assay | IC50=5.22 μM | 25761479 | |||
| SNU-175 | Growth Inhibition Assay | IC50=1.51 μM | 25761479 | |||
| COLO-320DM | Growth Inhibition Assay | IC50=5.38 μM | 25761479 | |||
| DLD-1 | Growth Inhibition Assay | IC50=8.65 μM | 25761479 | |||
| DiFi | Growth Inhibition Assay | IC50=10.95 μM | 25761479 | |||
| HCT-15 | Growth Inhibition Assay | IC50=8.64 μM | 25761479 | |||
| SCM-1 | Growth Inhibition Assay | IC50=17.5 μM | 25789057 | |||
| TMK-1 | Growth Inhibition Assay | IC50=22.6 μM | 25789057 | |||
| MKN-45 | Growth Inhibition Assay | IC50=14.0 μM | 25789057 | |||
| AGS | Growth Inhibition Assay | IC50=10.6 μM | 25789057 | |||
| S3 | Growth Inhibition Assay | IC50=53.5 ± 1.5 μM | 25801007 | |||
| SiHa | Growth Inhibition Assay | IC50=0.8 ± 0.1 μM | 25801007 | |||
| HT-29 | Growth Inhibition Assay | IC50=35.6 μM | 26003085 | |||
| DLD-1 | Growth Inhibition Assay | IC50=32.2 μM | 26003085 | |||
| HT29 | Growth Inhibition Assay | IC50=63 μM ± 18 | 26004084 | |||
| MC38 | Growth Inhibition Assay | IC50=23 μM ± 2 | 26004084 | |||
| SW620 | Growth Inhibition Assay | IC50=3.68 μM | 26023085 | |||
| SW480 | Growth Inhibition Assay | IC50=2.86 μM | 26023085 | |||
| RKO | Growth Inhibition Assay | IC50=1.23 μM | 26023085 | |||
| LoVo | Growth Inhibition Assay | IC50=1.2 μM | 26023085 | |||
| KM12 | Growth Inhibition Assay | IC50=4.37 μM | 26023085 | |||
| HCT116p53- | Growth Inhibition Assay | IC50=1.08 μM | 26023085 | |||
| HCT116 | Growth Inhibition Assay | IC50=1.04 μM | 26023085 | |||
| HCT15 | Growth Inhibition Assay | IC50=1.43 μM | 26023085 | |||
| HT29 | Growth Inhibition Assay | IC50=2.69 μM | 26023085 | |||
| DLD1 | Growth Inhibition Assay | IC50=2.01 μM | 26023085 | |||
| Colo205 | Growth Inhibition Assay | IC50=3.33 μM | 26023085 | |||
| BE | Growth Inhibition Assay | IC50=3.33 μM | 26023085 | |||
| CT26 | Cell Viability Assay | 4 mM | 48 h | decreases cell viability to 53.2% | 26137012 | |
| CT26 | Function Assay | 4 mM | 48 h | increases the expression levels of autophagy-related proteins, such as LC3-II, Beclin1 and ATG5 | 26137012 | |
| CT26 | Function Assay | 4 mM | 48 h | induces autophagy | 26137012 | |
| SK-OV-3 | Function Assay | 50 μM | 48 h | promotes sensitivity of ovarian carcinoma to NK cell-mediated cytolysis | 26138671 | |
| OVCAR-5 | Function Assay | 20 μM | 24h | promotes sensitivity of ovarian carcinoma to NK cell-mediated cytolysis | 26138671 | |
| PA-1 | Function Assay | 10 μM | 24h | promotes sensitivity of ovarian carcinoma to NK cell-mediated cytolysis | 26138671 | |
| SK-OV-3 | Function Assay | 50 μM | 96 h | up-regulates the stress ligands for NK cell-activating receptors and TRAIL receptors | 26138671 | |
| OVCAR-5 | Function Assay | 30 μM | 48h | up-regulates the stress ligands for NK cell-activating receptors and TRAIL receptors | 26138671 | |
| PA-1 | Function Assay | 10 μM | 48h | up-regulates the stress ligands for NK cell-activating receptors and TRAIL receptors | 26138671 | |
| SK-OV-3 | Function Assay | 50 μM | 96 h | triggeres the production of type I IFNs and chemokines | 26138671 | |
| OVCAR-5 | Function Assay | 30 μM | 48h | triggeres the production of type I IFNs and chemokines | 26138671 | |
| PA-1 | Function Assay | 10 μM | 24h | triggeres the production of type I IFNs and chemokines | 26138671 | |
| SK-OV-3 | Cell Viability Assay | 0-100 μM | 24/48/72 h | inhibits cell viability in both time and dose dependent manner | 26138671 | |
| OVCAR-5 | Cell Viability Assay | 0-60 μM | 24/48/72 h | inhibits cell viability in both time and dose dependent manner | 26138671 | |
| PA-1 | Cell Viability Assay | 0-20 μM | 24/48 h | inhibits cell viability in both time and dose dependent manner | 26138671 | |
| HCT116 | Growth Inhibition Assay | IC50=0.41 ± 0.02 μM | 26148596 | |||
| HT29 | Growth Inhibition Assay | IC50=0.88 ± 0.2 μM | 26148596 | |||
| SNU-387 | Growth Inhibition Assay | IC50=25 ± 2.7 μM | 26160429 | |||
| SNU-475 | Growth Inhibition Assay | IC50>30 μM | 26160429 | |||
| Hep-G2 | Growth Inhibition Assay | IC50=13.1 ± 1.6 μM | 26160429 | |||
| SNU-398 | Growth Inhibition Assay | IC50=6.5 ± 1.1 μM | 26160429 | |||
| LoVo | Function Assay | 1/5 μM | 24/48 h | induces transcriptional repression of DUT-N | 26208523 | |
| HCT116 p53+/+ | Function Assay | 1/5 μM | 24/48 h | induces transcriptional repression of DUT-N | 26208523 | |
| MDA-MB-231 | Growth Inhibition Assay | IC50=23.1 ± 0.1 μM | 26211591 | |||
| MCF-7 | Growth Inhibition Assay | IC50=15.4 ± 0.3 μM | 26211591 | |||
| SK-BR-3 | Growth Inhibition Assay | IC50=31.0 ± 0.1 μM | 26211591 | |||
| LoVo | Growth Inhibition Assay | 48 h | IC50=94.83 μM | 26269759 | ||
| HCT116 | Growth Inhibition Assay | 48 h | IC50=11.86 μM | 26269759 | ||
| SW480 | Growth Inhibition Assay | 48 h | IC50=1.87 μM | 26269759 | ||
| CaES-17 | Cytotoxicity Assay | 0–160 μM | 48 h | IC50=5.5 ± 0.2 μM | 26474693 | |
| HKESC-2 | Cytotoxicity Assay | 0–160 μM | 48 h | IC50=5.8 ± 0.5 μM | 26474693 | |
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | Oxaliplatin is a DNA alkylating agent that activates autophagy. Oxaliplatin inhibits DNA synthesis by conforming DNA adducts in RT4, TCCSUP, A2780, HT-29, U-373MG, U-87MG, SK-MEL-2, and HT-144 cells.Solutions are unstable and should be fresh-prepared.DMSO is not recommended to dissolve platinum-based drugs, which can easily lead to drug inactivation. | |
|---|---|---|
| Features | This product is not recommended to be dissolved in dimethylsulfoxide (DMSO).[9] | |
| Targets |
|
| In vitro | ||||
| In vitro | The main mechanism of action of Oxaliplatin is mediated through the formation of DNA–adducts. Oxaliplatin induces primary and secondary DNA lesions that lead to cell apoptosis. [1] Oxaliplatin is active against human melanoma cell lines C32 and G361 with IC50 of 0.98 mM and 0.14 mM, respectively. [2] Oxaliplatin effectively inhibits bladder carcinoma cell lines RT4 and TCCSUP, ovarian carcinoma cell line A2780, colon carcinoma cell line HT-29, glioblastoma cell lines U-373MG and U-87MG, and melanoma cell lines SK-MEL-2 and HT-144 with IC50 of 11 μM, 15 μM, 0.17 μM, 0.97 μM, 2.95 μM, 17.6 μM, 30.9 μM and 7.85 μM, respectively. [4] |
|||
|---|---|---|---|---|
| Cell Research | Cell lines | RT4, TCCSUP, A2780, HT-29, U-373MG, U-87MG, SK-MEL-2 and HT-144 cell lines | ||
| Concentrations | ~100 μM | |||
| Incubation Time | 48 hours | |||
| Method | The cytotoxicity studies are carried out with the sulforhodamine-B microculture colorimetrie assay. Typically, cells are plated into 96-well plates on day 0 and exposed to Oxaliplatin on day 1; the sulforhodamine-B assay is carried out 48 h after Oxaliplatin exposure. The plates are incubated at 37 °C in 5% CO2 and 100% relative humidity at all times except when adding Oxaliplatin and during the final assay period. The initial number of cells plated for the assay ranged from 2-20 × 103 cells/50 /nL/well. The numbers of cells for plating and the drug exposure time are based on pilot studies using the criteria that (a) the cells in control wells are still in the log phase of growth on the day of the assay; (b) the maximum absorbance for the untreated controls on the day of the assay is in the range of 1.0 to 1.5; and (c) cells go through >2 doublings during the drug exposure. Eight wells are used per concentration. The plates are read at 570 and/or 540 nm using a Biotek Instruments model EL309 microplate reader interfaced with an IBM PC-compatible computer. The data are transferred and transformed into a LOTUS 1-2-3 format by the computer program DATALOG, and survival fractions are calculated by comparing the drug treated with control |
|||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | p-Src(Y418) / p-FAK(Y861) p-AKT(Ser473) / AKT / PTEN / p-Src(Tyr416) VEGFR-1 / NRP-1 |
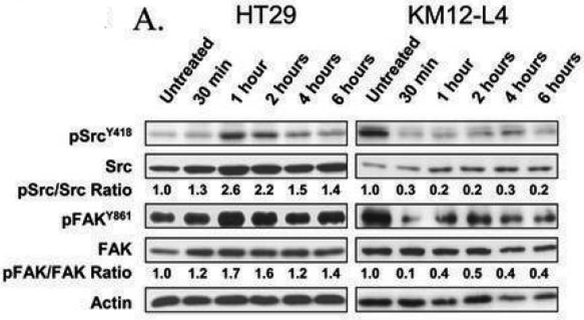
|
19383922 | |
| Immunofluorescence | ATXN2L / G3BP1 E-cadherin / Vimentin |
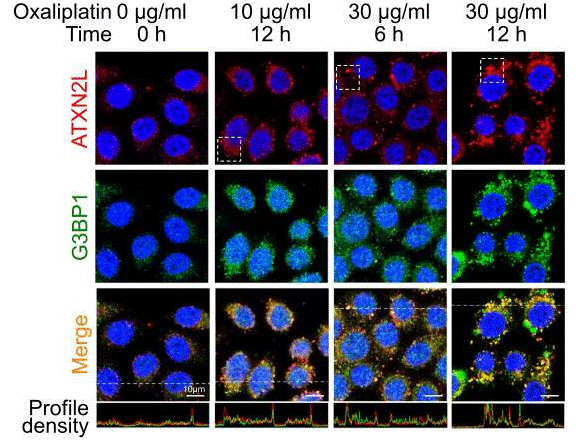
|
30787271 | |
| Growth inhibition assay | Cell viability |
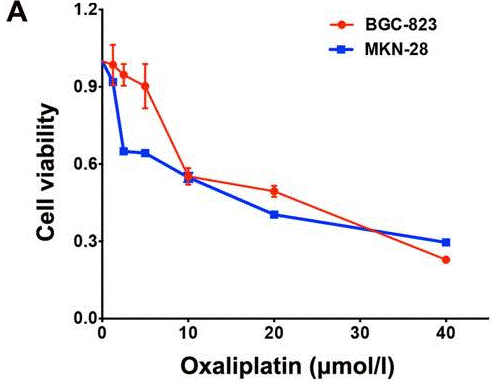
|
28339092 | |
| In Vivo | ||
| In vivo |
A weekly i.p. injection of Oxaliplatin at 10 mg/kg to nude mice bearing hepatocellular HCCLM3 tumors significantly reduces tumor volume and apoptotic index. [6] Oxaliplatin (5mg/kg, i.v. on days 1, 5 and 9) is active on T-leukemia-lymphoma L40 AKR with T/C of 1.77. Oxaliplatin is also efficient on intracerebrally grafted L1210 leukemia, MA 16-C xenografts, B16 melanoma xenografts, Lewis lung xenografts and C26 colon carcinoma xenografts. [7] Oxaliplatin induces impairment of retrograde neuronal transport in mice. [8] |
|
|---|---|---|
| Animal Research | Animal Models | Human hepatocellular carcinoma xenografts HCCLM3 |
| Dosages | 10 mg/kg | |
| Administration | A weekly i.p. injection | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05922358 | Not yet recruiting | Gastrointestinal Tumors |
Fujian Cancer Hospital |
September 1 2023 | Phase 2 |
| NCT05780684 | Recruiting | Colorectal Cancer|Esophagus Cancer|Appendix Cancer|Small Bowel Cancer|Ampullary Cancer |
Dartmouth-Hitchcock Medical Center |
July 14 2023 | Not Applicable |
| NCT05322590 | Recruiting | Metastatic Colorectal Carcinoma|Neuropathy |
Bexion Pharmaceuticals Inc.|ICON plc|CTI Clinical Trial and Consulting Services |
January 9 2023 | Phase 1|Phase 2 |
| NCT05068531 | Recruiting | Colorectal Cancer Metastatic |
Centre hospitalier de l''Université de Montréal (CHUM)|Exactis Innovation|Chaire Roger Des Groseillers d''oncologie chirurgicale HBP de l''Université de Montréal|Canexia Health |
September 1 2022 | -- |
| NCT05031975 | Recruiting | Colorectal Cancer |
Fondazione IRCCS Istituto Nazionale dei Tumori Milano |
May 2 2022 | Phase 2 |
| NCT05025826 | Recruiting | Metastatic Gastric Cancer |
Nantes University Hospital|Algosource |
April 1 2022 | Not Applicable |
Chemical Information & Solubility
| Molecular Weight | 397.29 | Formula | C8H14N2O4Pt |
| CAS No. | 61825-94-3 | SDF | Download Oxaliplatin SDF |
| Smiles | C1CCC(C(C1)[NH-])[NH-].C(=O)(C(=O)O)O.[Pt+2] | ||
| Storage (From the date of receipt) | 2 years 4°C(in the dark) powder | Solutions are unstable. Prepare fresh or purchase small, pre-packaged sizes. Repackage upon receipt. | |
|
In vitro |
Water : 2 mg/mL Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Frequently Asked Questions
Question 1:
Is it ok to dissolve Oxaliplatin in saline?
Answer:
Oxaliplatin is partially converted to cisplatin and L-isomers when dissolved in saline. The L-isomer of oxaliplatin is inactive. DMF is a better choice.
Question 2:
Is it ok to dissolve Oxaliplatin in DMSO?
Answer:
Even though cis-platin is soluble in DMSO, the use of DMSO to dissolve cis– or trans-diamminedichloroplatinum (DDP) in biological studies is strongly discouraged. The DMSO inserts itself into the ligand and inactivates platin-containing compounds. DMF is a much better choice than DMSO.
Tags: buy Oxaliplatin | Oxaliplatin supplier | purchase Oxaliplatin | Oxaliplatin cost | Oxaliplatin manufacturer | order Oxaliplatin | Oxaliplatin distributor